In this experiment we put our new patented prototype TEM holder to the test by exploring the process of Calcium Carbonate (CaCO₃) precipitation as a supersaturated solution inside the Nano Channel Chip was heating up.
The standout feature of this new type of TEM holder is its seamlessly integrated electrical circuit, which brings together a temperature sensor and resistor in the tip of the holder, for precise thermal control of the entire chip, tip, and all the liquid inside. By leveraging the well-known relationship between temperature and current, T=I^2⋅R, the system dynamically adjusts the current based on real-time feedback from the temperature sensor, ensuring accurate and responsive heating.
This allows for the system to be very user-friendly, as the heat applied to the system can be fine-tuned by simply turning a small control wheel (with no complex programming necessary).
In this experiment, we set out to examine how temperature affects the solubility of CaCO₃ water. As the temperature increases, by applying more current to the resistor, the super-saturated CaCO3 solution began to precipitate. This behaviour is grounded in thermodynamics and aligns with the principle that CaCO₃’s solubility will decrease as the temperature is increased.
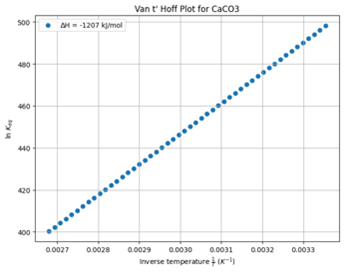
This behaviour is also supported by the fact that the formation of CaCO₃ is an exothermic process, meaning it releases heat. According to the Van ‘t Hoff equation, this results in an inverse relationship between solubility and temperature, as described by the positive slope in its Van ‘t Hoff plot (as seen below).
Before starting the temperature ramp-up, we carefully filled the nanochannels with the supersaturated CaCO₃ solution, as illustrated in the images below. Initially, the channels appeared uniform and featureless with minimal visible activity. At this stage, no crystals were present, and the uniform appearance served as a clear baseline, indicating our system was ready for temperature induced crystallization. This initial state can be clearly observed in the accompanying video below.


As the temperature was gradually increased towards the target of 80°C, the previously clear solution within the nanochannels began to transform dramatically. New, distinct features emerged, marking the onset of CaCO₃ crystallization.

The formation of these crystals was further confirmed by diffraction analysis. Diffraction patterns collected at the stable temperature of 80°C revealed sharp, well-defined spots, characteristic of crystalline structures.
